If we could only get the beta cells in the pancreas to rejuvenate themselves, we could then cure diabetes. The problem also involves shutting off immune responses, and eliminating behavioral and environmental factors that precipitate diabetes.
When there is an immune response that attacks beta cells, we have Type 1 diabetes. Over a fairly short period of time, it results in complete destruction of insulin-producing beta cells in the pancreas.
If we don’t remove the cells from their high sugar environment, and continue to damage them by subjecting them to high sugar, we run the risk of tipping to Pre-Diabetes, or Type 2 Diabetes.
We need to continually nurture and care for our beta cells, by making sure their environment is not high in sugar, and that they do not have to work too hard. If the beta cells have to work too hard due to high sugar, they will eventually give out. Our beta cells can only take so much.
Contents
What are beta cells?
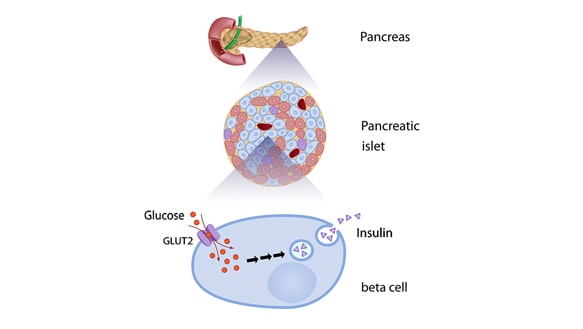
Beta cells are cells that are located in the pancreas, within the Islets of Langerhans. The Islets of Langerhans are in an area of the pancreas that serves to regulate endocrine function, by secreting hormones. The pancreas is involved in secreting insulin, glucagon, and some other hormones.
Beta cells functioning
The beta cells are little insulin producing machines. They manufacture, store, and release insulin via the pancreas. Without their full function, we can develop conditions such as Pre-Diabetes and Type 2 Diabetes.
In pancreas where there is no Pre-Diabetes or Type 2 Diabetes process going on, the beta cells respond extremely quickly to blood sugar changes. The beta cells will release some insulin that they have stored, and begin to make more insulin in response to rising blood sugar.
In a person with no diabetes, it takes about ten minutes for the beta cells to release stored insulin, and manufacture new insulin to take care of the high blood sugar. As you can see, this tends to wear out a tiny machine. It’s a hard job to do this all day long.
I highly recommend reading the following articles:
The importance of beta cells
Without beta cells in the pancreas, we are unable to make insulin. Without insulin, we can’t deal with the sugar in our blood that comes from the food that we eat.
If the sugar builds up, it clogs our system of veins and arteries, large and small. When our circulatory system gets clogged up with sweet, sticky blood, we get problems everywhere in our body. From the tiniest veins in our eyes and toes, to the largest arteries serving our heart and brain, high blood sugar takes a toll on our body.
That’s why taking care of our beta cells should be as important as taking care of our teeth, our eyes, or even our heart. Even though we can’t see them, the beta cells serve a grand function in the human body. Without them, things run amok. With their absence in our body, we turn to get our insulin from outside the body.
These tiny beta cells are the generals in our body, giving orders and supplies to bring down blood sugar. If they leave the battlefield, battle strategy immediately starts to fray. We are unable to battle against the bombardment of high blood sugar. Eventually, high blood sugar wins the battle and the generals are defeated.
Outside forces have to come in to save the day. This means oral medications for Type 2 Diabetes, followed by insulins (injected, inhaled, or pumped) if things get worse. This is when the importance of beta cells becomes inherently clear.
What is the role of beta cells in diabetes?
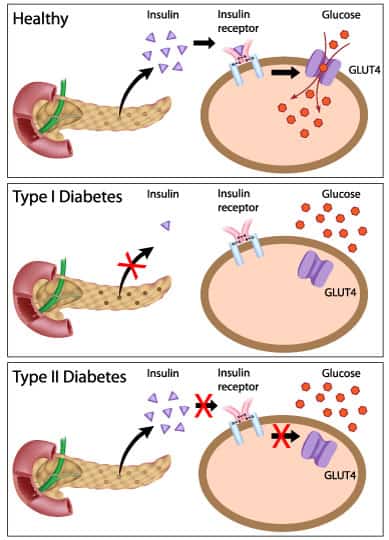
Beta cells in diabetes are either absent, damaged, or lost. Having Type 1 or Type 2 Diabetes makes a difference in whether or not your beta cells function.
Let’s look at the difference.
Type 1 diabetes
In Type 1 diabetes, beta cells are lost, not to be regained (at least there is no cure yet). There is an autoimmune process that occurs, where the body attacks the beta cells in the pancreas. It’s not a slight attack, but more like a full-on annihilation of beta cells that occurs.
Think Star Wars. The enemy comes in, and all the good guys get blasted out.
It is through stress induced at the cellular level that the beta cells are unduly attacked by the body. The exact cause of Type 1 Diabetes remains unknown. It is incurable, and a person must take insulin to survive.
To find a cure for Type 1 Diabetes, we need to first find out why the body attacks itself, and stop the auto-immune response. Then we will need to somehow rejuvenate the beta cells in the pancreas. Islet Cell Transplants have been done, but without a mechanism to turn off the immune response, all efforts at rejuvenation are futile. The immune system attacks the new beta cells again.
Type 1 Diabetes has a genetic component, and runs in families. I ran into a person with Type 1 Diabetes who was diagnosed following treatment with chemotherapy and radiation for prostate cancer at age 60. Another patient was a farm worker who became Type 1 in his middle ages. There are also environmental reasons for getting diabetes and research is still looking into that.
Type 2 diabetes
In Type 2 Diabetes, as we become more inactive, eat all the wrong things, and gain some weight, our bodies can become what are called “insulin resistant.” When our body is not paying attention to our insulin, and our cells hear our insulin knocking, but it can’t seem to come in, that is when our beta cells try to produce more insulin. This can also happen in youth who are obese, called MODY (Mature Onset of Diabetes in Youth).
The beta cells are signaled to release more stored insulin, and produce more insulin. Due to overworking, your beta cells get tired and start malfunctioning. They are no longer capable of doing their job. Something breaks in them. They can be going at 150 to 450 percent of capacity when you are on a sugar binge.
As a CDE friend of mine used to say of beta cells, “they poop out.” The problem is that before things get better, they start to get worse. Hunger from high blood sugars causes the person in the beginning phases of insulin resistance to eat more, not less.
This further compounds problems arising from high blood sugar. The excess glucose over time is compounded by an increase in blood lipids, and proteins that are involved in the immune system. With this increased stress placed on beta cells, over time, and at a much more gradual rate than with Type 1 Diabetes, beta cell function is diminished.
Insulin resistance is also perpetuated by increased fat build up around the pancreas, or “visceral” fat, and is further compounded by a fatty liver. The insulin that you are making can’t be used well, so your beta cells make more and more.
They finally just give out because they are damaged, and in need of being removed from an environment of sugar to recover. If they are not removed from the sugar environment, they will die.
Other than these mechanisms, the exact cause of Type 2 Diabetes remains unknown. Type 2 Diabetes is somewhat reversible through a healthy diet, active lifestyle, and moderate weight loss, though the preferred term is “remission”.
In order to continue to have adequately functioning beta cells, you have to stop drowning them in sugar and fat.
That’s why once you reverse your Type 2 Diabetes, you can’t go back to your old habits and behaviors and expect to have your beta cells cooperate. They will just throw up the “Surrender” flag again.
What about Pre-Diabetes?
Even in a Pre-Diabetes state, it is estimated that we have decreased beta cell functioning by up to 20-30%. When blood sugars are not to a Type 2 Diabetes state yet, we are still often experiencing high blood pressure, high cholesterol, and mildly increased blood sugars that are affecting our arteries and veins throughout our body.
Before we reach to Type 2 Diabetes levels, we have already done damage, and are twice as likely to have heart disease compared to someone without Pre-Diabetes. This is the warning sign, the alarm bell, and the sign you do not want to ignore.
Other than starting before Pre-Diabetes shows itself, this is the best time to begin protection of the beta cells. Through lifestyle modifications and through oral medication such as metformin, being involved in a program, such as the National Diabetes Prevention Program, or DPP, is one way to adopt lifestyle changes with continued support.
You can find a DPP program near you here: National Diabetes Prevention Program (DPP)
In general, all types of diabetes have a genetic component, although all diabetes is not genetic. It’s a complex disease, at best. Scientists are just beginning to understand the whole interworking of the human endocrine system. Only time will tell what they may find with the multitude of research that is being conducted today. In laboratories, diabetes has been cured in mice, but it has proved to be a much more difficult task to cure diabetes in humans.
What hormones do the beta cells secrete?
Just when you thought that beta cells didn’t have enough to do, it turns out they are also busy making other hormones, too. The beta cells secrete the hormones Amylin and C-peptide.
- Amylin
Amylin is the short stop. It stops blood sugar, and makes it wait a while before it is allowed to enter the blood stream.
Amylin is made in the beta cells, along with C-peptide. It is another peptide hormone. People with diabetes have deficient amylin (Type 2), and absent amylin (Type 1). Amylin stops the sugar (glucagon) from being secreted, slows the gastric system, and curbs appetite.
- C-peptide
C-peptide gets into your blood stream at about the same rate as insulin does. Its purpose is to come along behind blood sugar in your veins and arteries, and sweep up the mess that high blood sugars leave behind. C-peptide will also patch up any damage caused by high blood sugar from high carbohydrate meals that has been consumed.
Think about that for a minute. If the beta cells are damaged or worse yet, completely non-functioning, then they can’t make C-peptide that repairs the damage, either.
How are beta cells destroyed?
Beta cells are destroyed either by a surprise immune attack, as in Type 1 Diabetes, or by a gradual beating up of the beta cells that produce insulin. If damage is swift and severe, Type 1 Diabetes ensues, and the person must take insulin for the rest of their life.
If damage is over time, and new lifestyle changes related to healthy eating, increased physical activity, and moderate weight loss are not adopted, beta cell function may halt. Once destroyed completely, there is no method to rejuvenate the beta cells.
Besides immune beta cell destruction in Type 1, and obesity and insulin resistance in Type 2, other ways that insulin-producing beta cells can be destroyed include inflammation, and excess saturated and Trans fats in the diet, whether overweight or not.
Before beta cells are completely destroyed, they go through a period of exhaustion. In other words, they get sick before they die.
Is beta cell destruction linked to Alzheimer’s disease?
Scientists are steadily working to find a way to rejuvenate beta cells, or to take other cells in the pancreas and morph them into cells that mimic beta cell function. It seems that beta cell rejuvenation in humans may be on the horizon in the near future.
Some scientists are studying how amyloid deposits in the brain, as found in Alzheimer’s disease, are similar to amyloid deposits that are found in the beta cells that cause cell destruction and death. These insulin-producing cells find amyloid deposits in the cell wall toxic, and this is one way in which beta cells are destroyed.
In a person without Alzheimer’s disease, or Type 2 Diabetes, amyloid formation is inhibited by unknown factors. At Yale University, researchers are trying to find out what causes amyloid deposits to build up in people with Type 2 Diabetes and Alzheimer’s disease. They are specifically trying to find out what it is about amyloid build-up that damages beta cells, causing Type 2 Diabetes.
In a controlled laboratory, scientists were able to show that the cells of people without Type 2 Diabetes or Alzheimer’s disease did not allow for the formation of amyloid deposits. The researchers concluded that there is something about these healthy beta cells that is not found in the beta cells of people with Type 2 Diabetes or Alzheimer’s disease.
The researchers also tried to cause damage to the beta cells by introducing amyloid fibers. The amyloid deposits alone were not enough, but when combined with a protein chain, they were poison to the beta cells. The cells were then damaged, and slowly died. Similarly, a beta peptide has been implicated as a protein connected to the amyloid deposit build-up in the brain of Alzheimer’s patients.
Can I preserve beta cells?
It appears that High Density Lipoproteins (HDL) have possibly beneficial effects on beta cells in the pancreas. HDL, or “good” fat tend to protect beta cells, by preventing stress reaction of the cell, and eventually beta cell death. HDL is also known to help aid in insulin secretion by beta cells.
In Type 2 Diabetes, where damage occurs over time, new lifestyle changes related to healthy eating, increased physical activity, and moderate weight loss can be adopted. Depletion of beta cell function may halt, or even improve.
It’s possibly that beta cell function returns to a near normal level with the right interventions. Once destroyed completely, there is no method to rejuvenate the beta cells that make insulin at present.
With all of the beta cell protection you may be getting with HDL “good” fats, I see a trip to the grocery store for extra virgin olive oil, avocados, nuts and seeds, and maybe some almond milk. An old fashioned prescription for a healthy heart, diabetes friendly diet, lots of enjoyable physical activity, and some moderate weight loss should go a long way to preserve beta cells, since we know that damage occurs when we don’t lead a healthy life.
Can damaged beta cells be repaired?
What about fixing the damaged beta cells? Is there any hope for beta cells when they are all tired out, and they have no desire or ability to produce insulin anymore? Is there a way to suddenly wake them up, and have them spring into new life?
When there is no autoimmune attack, as in Type 1 Diabetes, and high blood sugar is no longer present, beta cells can rejuvenate themselves. They all replicate, just like other cells in your body. In mice, beta cells multiply between 2% and 15%, which shows a high rate of variability from mouse to mouse for the ability to replicate these important cells.
For the beta cells that do replicate, they divide every 12 to 20 days. When the metabolic rate is altered, beta cells may not replicate as much due to increased insulin demand. In other words, they swap one job out to do another.
Human beta cells live longer than rodent cells. Our beta cells replicate the most from birth through five years. Then it starts to taper off, with the least beta cell replication occurring in the elderly population.
Can we repair or replenish beta cells by medication or naturally?
It is not possible with Type 1 Diabetes. However, with Type 2 Diabetes, metformin and some other diabetes medications may serve to bring down the high blood sugar, and protect the beta cells from further destruction. Once the beta cells are let out of their high sugar environment, they are then able to repair themselves, and replicate.
Can I increase my beta cells?
Yes, by eating fats, such as olive oil and seed oils instead of butter or margarine, avocado, nuts and seeds, almond milk, and other fats considered to have HDL, or “good fats, you can increase your beta cells. Exercise and weight loss, and keeping your blood sugar in a target range will help to keep your beta cells out of a high blood sugar environment.
They tend to recover from damage, but one must take swift action before they are completely destroyed. Get your biomarkers in range, and beta cells should start to multiply again.
What are the lifespan of beta cells?
In mice, the half-life of beta cells is estimated at 30 days. Human beta cells seem to have a longer half-life, but the life span of human beta cells is unknown. Proliferation is greatest up to age 20.
What is the difference between beta and alpha cells?
Alpha cells, like beta cells, are located within the pancreas, the pancreatic islets. They are about 20% responsible for producing and secreting glucagon. Glucagon is another peptide hormone which serves to increase sugar in blood when it is low. Glucagon is also produced in the liver.
How is obesity related to the destruction of beta cells?
Obesity is considered to be a state of inflammation. Fat tissue accounts for much of the inflammation that occurs in obese people. Obesity is a risk factor for Pre-Diabetes and Type 2 Diabetes. Fat tissue is not sensitive to insulin, and a high saturated and Trans fat diet is associated with increased insulin resistance and beta cell damage.
Further reading:
Can a healthy diet and exercise improve beta cell functioning?
Absolutely, you can protect your beta cells by eating a heart healthy and diabetes friendly diet, low in carbohydrates and saturated/Trans fats. Eating from all food groups is important, and nutrients from our recommended daily vitamins help to keep the beta cells healthy. Insulin sensitivity is improved, and insulin resistance is lowered when we get regular physical activity. Weight loss will further enhance beta cell health.
We hope that this article has shed some light into the complex world of Beta cells and their importance in our body to keep diabetes at bay. However, if you have type 2 diabetes and do not want the total destruction of your beta cells, adopting a healthy and active lifestyle can increase their lifespan and help you in the long run. Please leave us any comments you may have. We love to hear from our readers.
TheDiabetesCouncil Article | Reviewed by Dr. Sergii Vasyliuk MD on May 27, 2020
References:
- http://www.diabetes.co.uk/body/beta-cells.html
- http://www.joslin.org/the-problem-of-not-enough-beta-cells-in-type-1-dabetes.html
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3608918/
- http://www.diabetes.org/research-and-practice/we-are-research-leaders/recent-advances/study-uncovers-new-ways-to-prevent-beta-cell-death.html/